Our Specification Management Platform
Elevate your team and supplier collaboration with an all-in-one Specification Management platform
Our patented, cloud-based Specification Data Management™ (SDM) platform enables you to manage your specs in one place, ensuring data accuracy and enabling collaboration across teams & suppliers.
Trusted by C-Suite

“What we’re doing with Specright is strengthening up the whole product life cycle management process. We have some island systems today that do some of these things, but all of those challenges – disconnected systems, arguments about data… these things just don’t make sense now. With Specright, we decided to put this PLM system in place and actually drive it forward.”

Frank Ardite
Global Quality & Digitization Director, BASF

“We’ve created a technology ecosystem where I’m not waiting on technology. And that’s why I really like Specright. That’s why I’m here is because they’re great partners when it comes to that. We work together.”

Aron Kolosik
Chief Supply Chain Officer, Central Garden & Pet
“Expanding our partnership with Specright to manage specification data across the entire product life cycle was an easy choice and sets us up for success in enhancing and developing innovative products while also ensuring our workforce is more efficient.”

Andy Norman
CIO, Bob Evans Farms
Our Architecture: A Secure & Stable Cloud-Based Platform Built for Specs
Our data architecture is centered around and optimized for specs, making the data in our platform more searchable, traceable, and ultimately more usable for your business. Cloud-based means both internal and external users have immediate , 24/7 access – anywhere in the world.
Specright is built on Force.com, the most scalable, reliable, and secure enterprise cloud platform in the world. All our Specright services are SOC2 compliant from security, confidentiality, and availability trust perspectives.
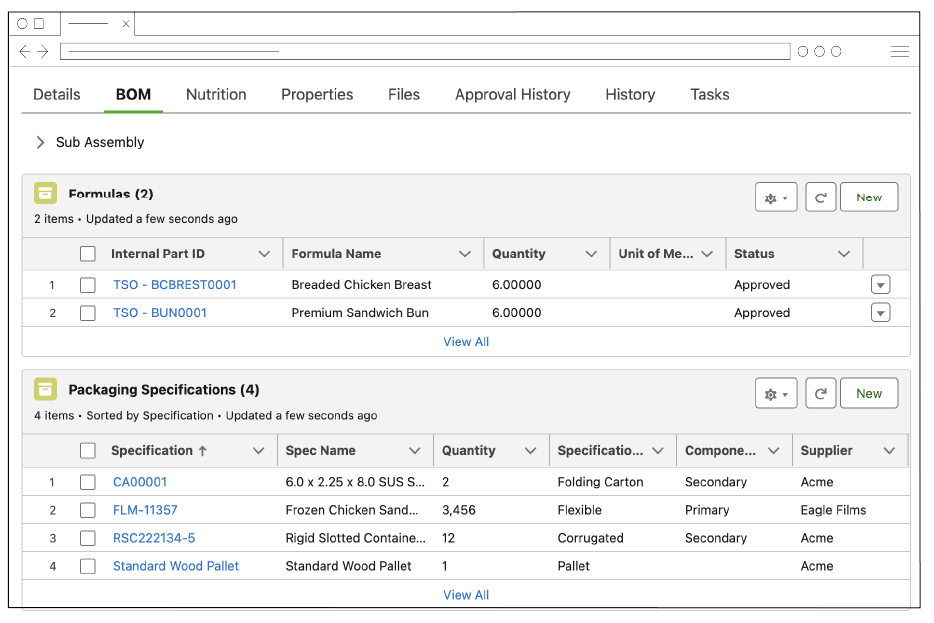
Streamline Your Product Development Process: One Platform for All Your Spec Needs
Digitize and standardize specifications, from raw materials, formulas, ingredients, packaging, products, and finished goods in one cloud-based platform. Easily link specs together to form sub-assemblies, bill of materials, and finished goods.
Key Features
- 150+ spec templates help you standardize and digitize data
- Universal search helps you find data in any spec or related document
- Build-in analytics flag duplicate specs and prevent SKU proliferation
- Version control, supersede, and history tracking for changes
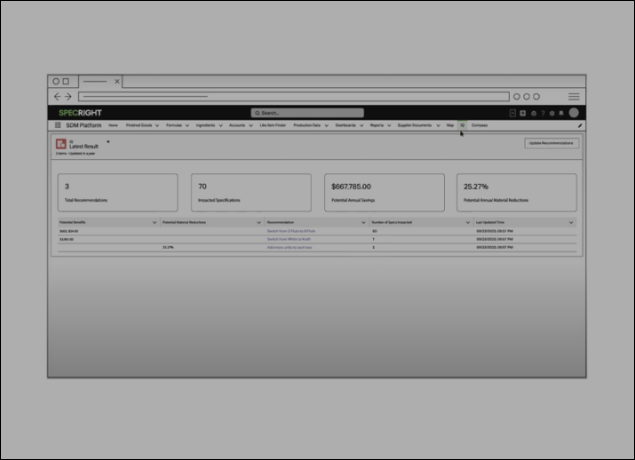
Transform Data into Action with Real-Time Recommendations
Specright IQ scans all of your existing specifications to help identify recommendations to help you save costs or drive sustainability.
Key Features
- Cost savings recommendations
- Sustainability recommendations
- SKU rationalization recommendations
2024 Gartner Market Guide: Packaging & Product Specification Management
Learn about:
- Regulatory Challenges: Regulatory requirements, legislation and costs of goods are exposing gaps and weaknesses in product and packaging specification data processes and practices.
- Sustainability Legislation: Growth in global sustainability legislation for manufacturers is exposing gaps in specification data management solutions, spec quality, completeness and accuracy that is forcing supply chain technology leaders and CIOs to seek solutions to meet compliance requirements.
- Outdated Processes: Many regardless of region, industry or size suffer from outdated or manual processes to manage specification data, resulting in growing strains on resources, cost overruns and risks of noncompliance.

Everything You Need in One Platform
Packaging Management
Our digital, searchable platform puts data at your fingertips – and keeps your packaging teams on the same page.
Product Data Management
Accelerate time to market with clean, up-to-date product data shared across teams and suppliers.
Supplier Collaboration
Bring suppliers into your spec-first PLM for faster collaboration with accurate, up-to-date specs and shared workflows.
New Product Development
Centralize specs, projects, and suppliers to accelerate speed-to-market.
Manufacturing Instructions
Link formulas, lines, processes, and process steps for clear, consistent execution.

Formulation, Labeling & Claims
Create and optimize formulas in one place, from concept to claims, with AI-driven automation and insights.
Artificial Intelligence
AI that actually works—because it’s built on accurate, spec-level data connected across the entire lifecycle.
Trusted by Fortune 500, private label retail & challenger brands











Easily Integrate with Your Existing Systems






2025 State of Specification Management Report
Companies are managing more complex specification data than ever—but outdated systems are holding them back. Our 2025 report reveals the top challenges facing packaging and supply chain teams today, from poor data accuracy and version control to mounting sustainability regulations.
Learn why 75% of respondents say they need better Specification Management, and how leading teams are transforming data chaos into competitive advantage.
Discover the Impact of Specification Management
Access our free resource library, and learn how to focus your data strategy, innovate, and drive your bottom line growth from industry experts.